sterna biologicals GmbH
company developing first-in-class, safe and
efficacious antisense oligonucleotide (ASO) based therapies targeting the transcription factor GATA-3. GATA-3 is a key master transcription factor with pivotal roles in cell development, (type 2) inflammation, and tumorigenesis. The Company’s proprietary DNAzyme based drug candidate hgd40 has shown therapeutic potential in four Phase IIa clinical trials. With this, sterna strives to develop GATA-3 specific novel immunomodulating therapies for millions of patients suffering from type 2 inflammatory and oncological diseases.
Sterna Biologicals at a glance:
| Field of Activity |
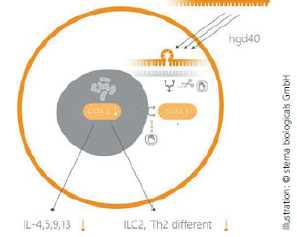
Sterna is the only company that has successfully made GATA-3 druggable. The Company has shown that downregulating GATA-3 leads to a reduced expression of key inflammatory cytokines, and thereby normalization of the dysregulated type 2 immunity (immunomodulation). In clinical trials, sterna has successfully demonstrated that GATA-3 downregulation could provide a novel, first-in-class treatment approach for a broad range of inflammatory and oncological diseases. |
| Ownership Structure and Financing | Privately held. |
| Products/Services |
With its proprietary DNAzyme hgd40 (a special type of antisense in various formulations), sterna has clinically demonstrated that downregulating GATA-3 allows the drug candidate to intervene with upstream inflammatory processes. Opposite to many existing treatments that only address specific individual inflammatory mediatory, hgd40 is expected to broadly antagonize several key inflammatory processes and mediators at the same time. The Company aims to exploit hgd40 and expand its potential in diseases with high unmet medical need, where downregulation of GATA-3 directly leads to clinical benefits for patients.
|
| Unique selling point | Hgd40 has several highly attractive characteristics:
• unmodified (natural) 34mer deoxynucleotide, stabilized by inverted thymidine at 3‘ end • cellular uptake without delivery system – specifically cleaves GATA-3 mRNA of humans and tox species – excellent safety profile; no immune system activation (e.g. via TLR-9) • fully automated solid phase manufacturing; well established, proven and robust chemical process technology; linear scalability into commercial scale • First-in-class: no on-target competition in clinical development |
| Date of Incorporation/ Number of Employees |
2006/14 |
Contact:

CEO and Managing Director
| Address | Bismarckstr. 7 35037 Marburg |
| Telephone/Telefax | +49 6421 98300-50/-599 |
| info@sterna-biologicals.com | |
| Web Address | www.sterna-biologicals.com |
| Social Media |
