A major theme for biopharma companies is the low rate of success in drug R&D, from early R&D through clinical development and regulatory approval. Moreover, from the patient’s and the physician’s perspective, drug R&D is a long path to wait for an effective treatment to be available. Around 85% of potential drugs entering Phase I clinical trials are destined to fail.1 By Robert Steininger
Even for successful drugs the path to approval is stacked with barricades: time consuming clinical trials, high regulatory standards and inordinately high costs. The R&D journey of new drugs that make it to market is about twelves years with average costs over EUR 1.3 bn phases.2
The ability to predict clinical trials outcomes at every stage would help to reduce the lack of efficiency inherent in drug R&D. A significant part of this inefficiency comes from “dirty” and scattered data, plus the inability to merge and map different semantics such as drug, clinical and molecular data and information.
Addressing these challenges creates a real opportunity for computer science and data management. With the latest developments in Artificial Intelligence plus Machine Learning (AI+ML) and data management there is a good chance the drug R&D world can be more efficient and less costly for patients and the entire healthcare ecosystem.
Molecular Health GmbH (Heidelberg, Germany) is focused on the capture, curation, integration, and analysis of large data sets in the fields of biomedicine, drugs and medical products. By combining these data sets with novel AI+ML technologies, precision medicine can be significantly advanced.
Precision medicine focuses on helping to prevent diseases and to find the best therapy based on individual disease conditions in each and every patient. In combination with computing technologies including AI, precision medicine holds great promise, for example, in AI-aided treatment decision support, drug development, outcomes-based medicine, value-based reimbursement and, importantly, AI-driven prediction of clinical trials probabilities of success.
Molecular Health has now developed MH Predict, a product to help address the R&D efficiency dilemma. MH Predict utilises AI by merging it with Molecular Health’s complete, comprehensive biomedical and drug knowledge database, Dataome. MH Predict is an application to predict clinical trials probabilities of success. It enables the improvement of clinical trial success and reduces clinical trial failure while optimizing resource allocation for the biopharma industry and investors and enhancing the overall efficiency of drug development.
The impact of better clinical trials success predictions is huge: lower attrition rates, optimized clinical trials sizes, shorter study duration, faster time-to-market and, thereby, longer patent protection. Importantly, this results in lower R&D costs, improved drug portfolio planning and higher returns on asset investments. In broad terms, the key benefits MH Predict provides are:
a) an accurate, up-to-date knowledge base,
b) the potential for greater success in resource and capital allocation, and
c) better drugs developed faster and at lower costs.
There are additional benefits. For example, MH Predict allows for greater accuracy in competitor analysis, which delivers better proprietary drug positioning, differentiation, pricing and marketing. Additionally, access to clean and accurate data will produce a much clearer understanding of M&A assets, better acquisition decisions and greater deal success.
Recently, MH Predict was officially released for users in the pharmaceutical and related industries. In addition, Molecular Health granted an exclusive license for the financial industries to DATEN CAPITAL LLP (London, UK), in January 2019. DATEN is a new firm run by Amit Karna, formerly an investment banker at Goldman Sachs, which will apply MH Predict to assist in investment decisions in the sector.
High Predictive Accuracy
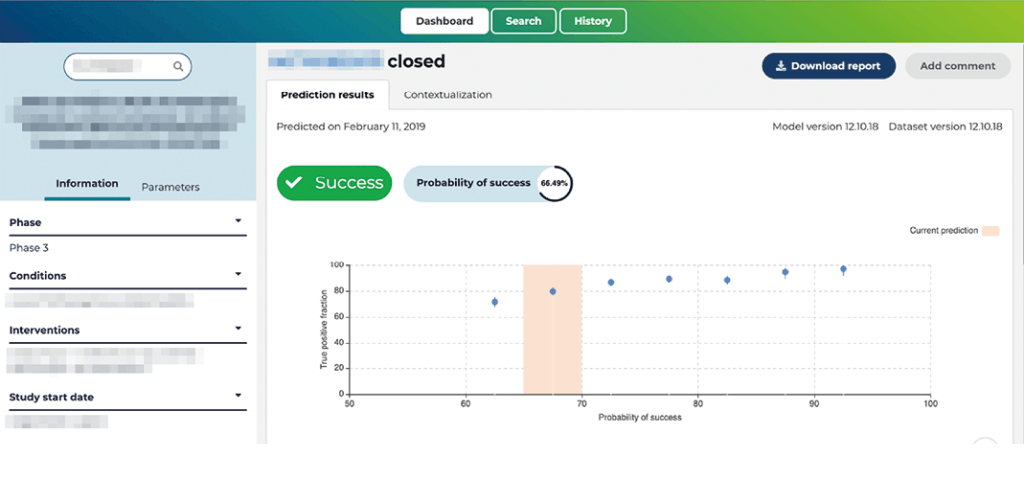
MH Predict is quality- and performance-tested by retrospective and prospective study data and challenges. A test on trials, in which the outcome was not known at the time of prediction, including trials at all clinical development phases, in different indications with small and large molecules, showed MH Predict to be 84% accurate. The product is now being launched as version 2.0, which robustly confirms its highly predictive accuracy.
“Our first goal at Molecular Health is to deliver AI models to the fingertips of biopharma R&D decision makers. These models we have developed enable R&D experts to predict clinical trial probability of technical success. With MH Predict, users are able to delve into the algorithms’ predictions,” said Blanca Baez, SVP, Global Head of Pharma & Biotech at Molecular Health.
The MH Predict screen (see fig. 2) is designed for clarity and simplicity of operation. In fact, it requires just three simple steps: enter the NCT identification code; click on “predict”; and read the reports.
Dataome
The real driver and differentiator of MH Predict is Molecular Health’s proprietary Dataome technology platform, a unique, high-quality curated, interoperable system that combines clinico-molecular and drug data with proprietary analytical processes. Molecular Health’s Dataome is one of the largest, most strictly curated clinico-molecular data repositories in the world and receives updates continuously as new data becomes available.
Dataome combines the curated data from public and private clinico-molecular sources, structured and unstructured, plus medical literature and randomized clinical trials with real-world data.
Dataome is unique in the information features it accesses, not just the standard drug, clinical trial and corporate data, but several further feature domains to ensure the algorithms access the latest information available to help enhance human decisions (see fig. 1: MH Predict). These additional domains enable Dataome to deliver over 300 feature domains for assessment by the ML algorithms in MH Predict.
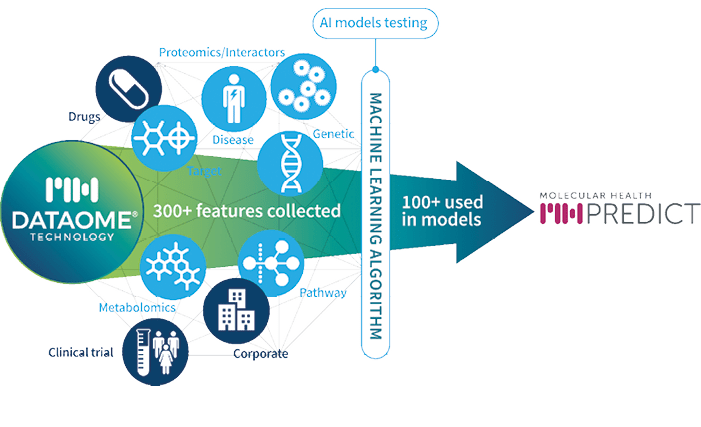
Dataome has taken over ten years to plan and build. Today the data features include highly curated data on important R&D domains, including, but not limited to: targets; diseases; genetics; pathways; proteomics/interactors; and metabolomics.
Based on Dataome, Molecular Health has developed applications, products, services and offerings for physicians, hospitals, laboratories, the pharmaceutical industry, regulators, payers and biopharma innovators.■
1) Smietana, Katarzyna & Siatkowski, Marcin & Møller, Martin. (2016). Trends in clinical success rates. Nature Reviews Drug Discovery. 15. 10.1038/nrd.2016.85.
2) Ingrid Torjesen (2015) Drug development: The journey of a medicine from lab to shelf. The Pharmaceutical Journal 12 MAY 2015.
Autor/Autorin

Robert Steininger
Robert Steininger ist Fachautor für u.a. Anlagestrategien und publiziert regelmäßig zu Fachthemen wie Online- und Investment-Strategien, Glücksspielthemen, Krypto und Verhaltensanalyse.
